|
Question 1008388: a bowl is made by cutting into half a hollow sphere of external diameter50.8cm, made of metal 2.54cm thick. if the bowl is completely filled with liquid of density 31.75kg/m^3 calculate the mass of liquid in grams.
Found 2 solutions by mananth, ikleyn:
Answer by mananth(16949)   (Show Source): (Show Source):
You can put this solution on YOUR website!
external diameter50.8cm, made of metal 2.54cm thick.
internal diameter = 50.80-2.54*2 = 45.76 cm = 0.4576 m
radius = 0.2288 m
Volume of hemisphere = 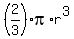
V = (2/3) *3.142 * (0.2288)^3
= 0.025 m^3
mass = d*v
mass = 0.025 * 31.75
=0.8255 kg
= 825.50 gms
Answer by ikleyn(53878)   (Show Source): (Show Source):
You can put this solution on YOUR website! .
a bowl is made by cutting into half a hollow sphere of external diameter50.8cm, made of metal 2.54cm thick.
if the bowl is completely filled with liquid of density 31.75kg/m^3 calculate the mass of liquid in grams.
~~~~~~~~~~~~~~~~~~~~~~~~
Calculations and the final answer in the post by @mananth are incorrect.
Below is my correct solution.
external diameter 50.8cm, made of metal 2.54cm thick.
internal diameter = 50.80-2.54*2 = 45.76 cm = 0.4576 m
internal radius = 0.2288 m
Volume of hemisphere = 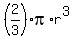
V = (2/3) * 3.142 * (0.2288)^3
= 0.025 m^3
mass = d*v
mass = 0.025 * 31.75
= 0.794 kg <<<---=== ANSWER
Solved correctly.
//////////////////////////////////
Been a curious person, I asked Google "which liquid has the density of 31.75 kg/m^3 ?"
Below is the Google answer.
There is no common pure liquid that has a density of 31.75 kg/m^3 at standard temperature and pressure.
This value is exceptionally low for a liquid; for comparison, water has a density of approximately
1.000 kg/m^3, and the lightest known liquid, liquid hydrogen, has a density of about 70.85 kg/m^3 at 20 K.
A density of 31.75 kg/m^3 is more characteristic of:
Gases under high pressure: Many gases reach this density when compressed significantly.
Supercritical fluids: Substances in a state between a gas and a liquid, such as carbon dioxide (CO2),
can be tuned to this specific density by adjusting temperature and pressure.
Foams or Aerogels: While these are solids, they are "liquid-like" in certain industrial contexts and can have
very low densities.
If this figure appeared in a specific textbook or problem set, it likely refers to a gas or a specific substance
at a non-standard, high-pressure state.
|
|
|
| |