Question 1126501: Pure acid is to be added to a 10% acid solution to obtain 90 L of 64% solution. What amounts of each should be used? How many liters of 100% pure acid should be used to make the solution?
Found 4 solutions by addingup, josgarithmetic, ikleyn, greenestamps:
Answer by addingup(3677)   (Show Source): (Show Source):
Answer by josgarithmetic(39799)   (Show Source): (Show Source):
Answer by ikleyn(53763)   (Show Source): (Show Source):
You can put this solution on YOUR website! .
Since both @addingup and @josgarithmetic gave incorrect solutions and wrong answers, I came to fix everything.
Let x be the volume of the pure acid to be added, in Liters.
Then the volume of the 10% acid should be 90-x.
Your equation is the balance of pure acid in both ingredients and in the mixture
pure acid + pure acid = total pure acid, or
1x + 0.1*(90-x) = 0.64*90.
Multiply by 100 both sides.
100x + 900 - 10x = 64*90
90x = 64*90 - 90
x = 64 - 10
x= 54 liters of the pure acid.
and 36 liters of the 10% acid.
Answer. 54 liters of the pure acid and 36 liters of the 10% acid.
Check. 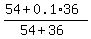 = 0.64 = 64% concentration. ! Correct ! = 0.64 = 64% concentration. ! Correct !
Solved.
-----------------
It is a standard and typical mixture problem.
For introductory lessons covering various types of mixture word problems see
- Mixture problems
- More Mixture problems
- Solving typical word problems on mixtures for solutions
- Typical word problems on mixtures from the archive
in this site.
You will find there ALL TYPICAL mixture problems with different methods of solutions,
explained at different levels of detalization, from very detailed to very short.
Read them and become an expert in solution mixture word problems.
Also, you have this free of charge online textbook in ALGEBRA-I in this site
- ALGEBRA-I - YOUR ONLINE TEXTBOOK.
The referred lessons are the part of this textbook in the section "Word problems" under the topic "Mixture problems".
Save the link to this online textbook together with its description
Free of charge online textbook in ALGEBRA-I
https://www.algebra.com/algebra/homework/quadratic/lessons/ALGEBRA-I-YOUR-ONLINE-TEXTBOOK.lesson
to your archive and use it when it is needed.
Answer by greenestamps(13334)   (Show Source): (Show Source):
You can put this solution on YOUR website!
Here is a quick and easy alternative to the standard algebraic method for solving mixture problems like this, as shown by the other tutors.
You are adding 100% pure acid to the 10% acid; the percentage of the mixture moves from 10% towards 100%. You stop when the percentage is 64%.
(1) 64% is 3/5 of the way from 10% to 100%. (Do a little mental arithmetic: 100-10 = 90; 64-10 = 54; 54/90 = 3/5)
(2) That means 3/5 of the mixture must be the 100% pure acid you are adding.
3/5 of 90 L is 54 L. So 54 L of the 100% acid and 36 L of the 10% acid.
|
|
|